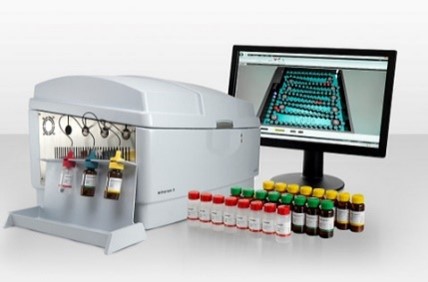
This growth-based sterility test detects microbial contamination based on the presence of microbial Adenosine Triphosphate (ATP) in a sample. Results are objective and based on instrument analysis. It works for filterable, nonfilterable, cloudy, and complex product formulations. The
test is non-destructive, enabling microbe identification for investigation. It has been validated and approved by international regulators and supports sterile product release at various pharmaceutical companies.