Lecture
Designing Material flow & Intralogistics in GMP environment
Lecture will be recorded!
Lecture will be recorded!
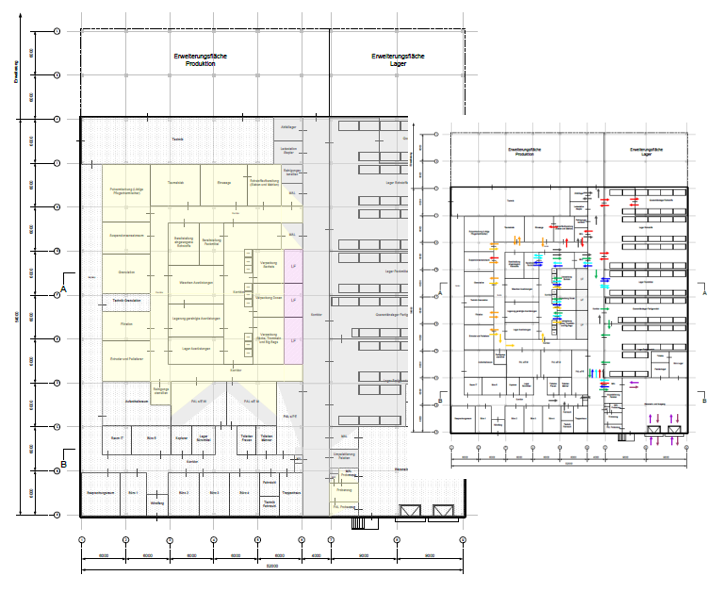
Planning material flow and intralogistics in GMP environments imposes strict requirements on layout design, particularly due to hygiene zones, airlocks, and the need to prevent cross-contamination. This presentation illustrates how these factors impact material flow and logistics and which solutions in the concept design phase help combine efficiency and compliance. Modular planning and automated transport systems enable flexible production areas but introduce specific intralogistics challenges. Real-world examples from pharma, food, and cosmetics demonstrate how regulatory demands and logistical needs can be successfully aligned.
Speakers:

Oliver Schubert
