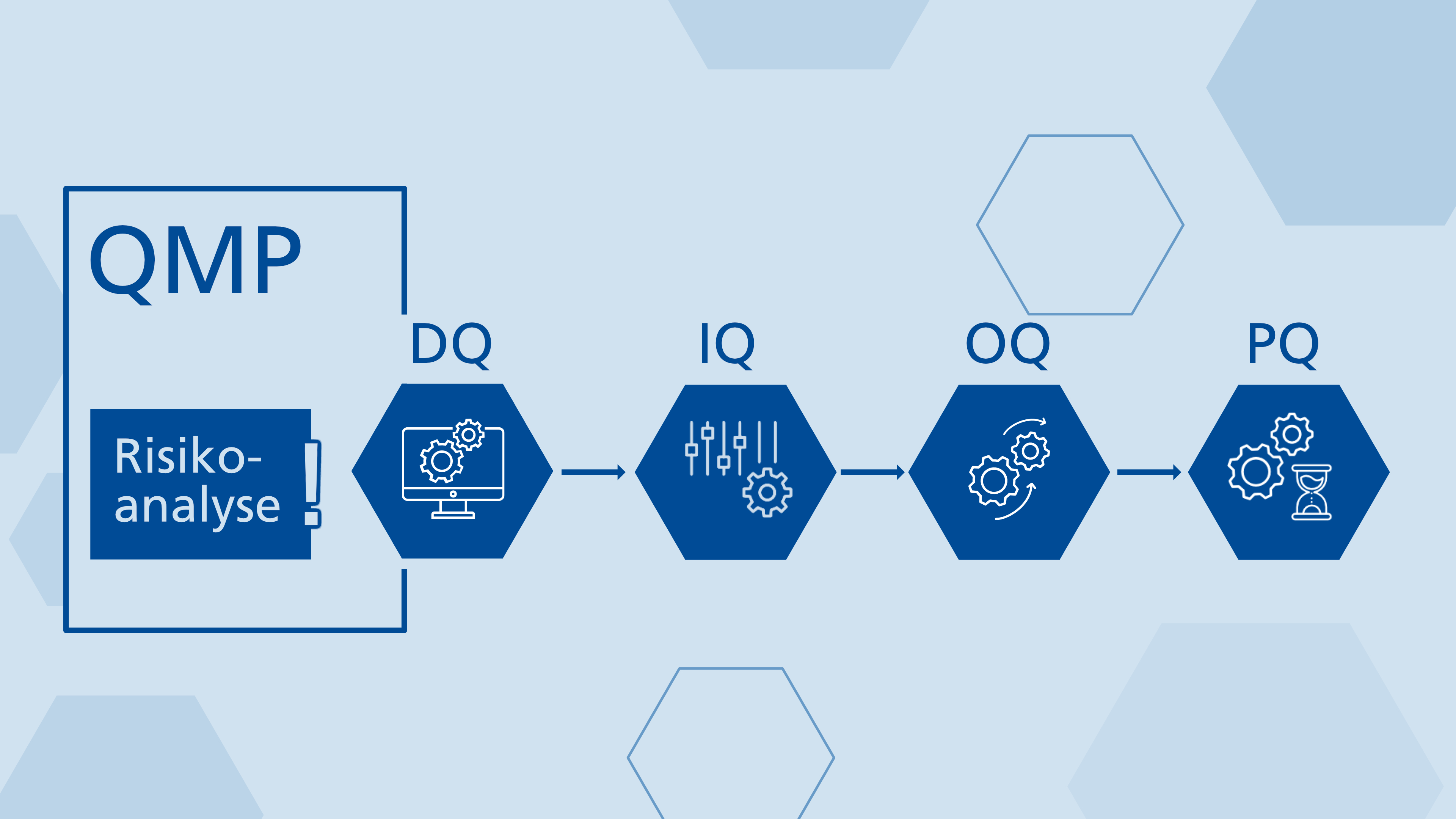
Qualifying technical systems as GMP-compliant is a crucial process in the pharmaceutical industry, clearly structured by the Qualification Master Plan (QMP). This article analyses the QMP as a central guide that defines responsibilities and procedures in the design qualification (DQ), installation qualification (IQ), operational qualification (OQ) and performance qualification (PQ) phases. A key element is the GMP risk analysis, which identifies risks and develops proactive measures to mitigate them. We discuss the critical importance of clearly delineating responsibilities between clients and suppliers, and of ensuring that employees have the necessary skills. We also emphasise the importance of thorough documentation to ensure traceability and compliance. Additionally, we highlight the qualification requirements from the authorities' perspective.

