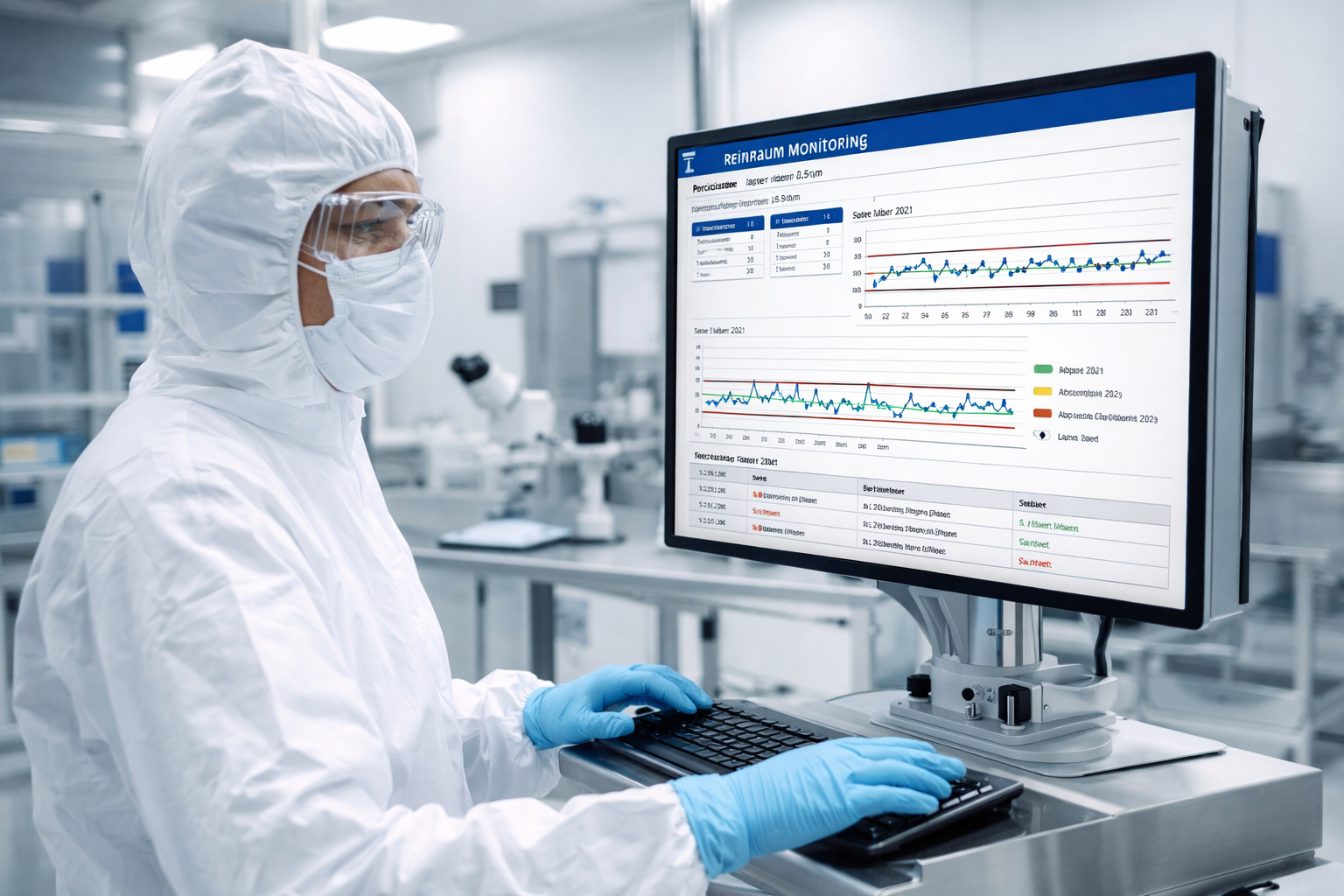
Because GMP requires not only individual measurements but also continuous verification that the cleanroom is under control, Annex 1 stipulates trend analyses.
Basic principle
Annex 1 aims to detect contamination at an early stage and permanently minimize the risk of contamination for sterile products. Individual measurement points (e.g., daily values) are not sufficient for this purpose, as gradual deterioration, seasonal effects, or systematic problems often only become apparent over time. Trend analyses identify patterns (increasing particle or germ counts, more frequent alarms, changes in germ flora) before alert or action limits are exceeded, enabling preventive measures to be taken. This approach is in line with the quality risk management and contamination control strategy (CCS) required in Annex 1, which calls for data-driven, risk-based control of cleanroom processes.
Formal anchoring of trending in Annex 1
The current version of Annex 1 defines that trend analyses of environmental and process monitoring data are necessary in order to detect system deterioration and contamination risks at an early stage. Particles, CFU, other room parameters, and alarm and fault messages from equipment and monitoring systems must be checked and evaluated in time series.
Continuous cleanroom monitoring vs. mobile cleanroom monitoring
Mobile monitoring methods (e.g., portable particle counters, active air samplers, contact samples) are also subject to trending requirements, as Annex 1 aligns all environmental monitoring—not just continuous systems—with a life cycle and trend-based approach. Data from portable devices and samples must be recorded and stored in such a way that they can be trended according to location, time, activity, and cleanroom status (at rest/in operation). This allows systematic identification of increasing germ counts at specific locations, recurring local hotspots, changes in germ flora, and differences between “at rest” and “in operation.” Annex 1 expects these trends to be incorporated into regular EM reviews and CCS assessments and to serve as a basis for adjusting sampling plans, limits, cleaning/disinfection, and training measures.


